Keratinocytes and Epidermis
The outer layer of the skin, the epidermis, is composed mainly by keratinocytes, which represent around 90% of the cells in that layer[1], [2]. They play a role in immune responses, by acting as barrier and preventing the entrance of bacteria, viruses, fungi and parasites[1]. Their defensive function also involves protection against water loss, heat and UV radiation[3].
The epidermis is subdivided in stratum basale, stratum spinosum, stratum granulosum, stratum lucidum and stratum corneum (Figure 1) [4]. These layers contain keratinocytes at different stages of the keratinisation process. The keratinisation process consists on a series of mitotic divisions that start at the stratum basale and that lead to the replacement of the cytoplasm with the protein keratin. At the end of the keratinisation process, keratinocytes can be found dead, as flat, nucleus-free and highly keratinised squamous cells at the stratum corneum[4].

Figure 1. Layers of the epidermis
Primary Normal Human Epidermal Keratinocytes (NHEK) kindly provided by PromoCell GmbH were cultured and observed under the 3D Cell Explorer.
Lamellar Bodies and Keratohyalin Granules
Keratinocytes at the stratum granulosum contain two characteristic structures that were visible in our footage: lamellar bodies and keratohyalin granules (Figure 2) [2].
While keratohyalin granules contain proteins involved in the aggregation of keratin filaments and in the formation of the cell envelope, lamellar bodies contain lipids and fuse with the plasma membrane in order to secrete the content into the extracellular space.
Regarding their size, keratohyalin granules are generally bigger (up to several micrometres)[5] than lamellar bodies (200-300nm)[6]. The 3D Cell Explorer allows us to visualise structures bigger than 200nm, thus making both structures distinguishable.
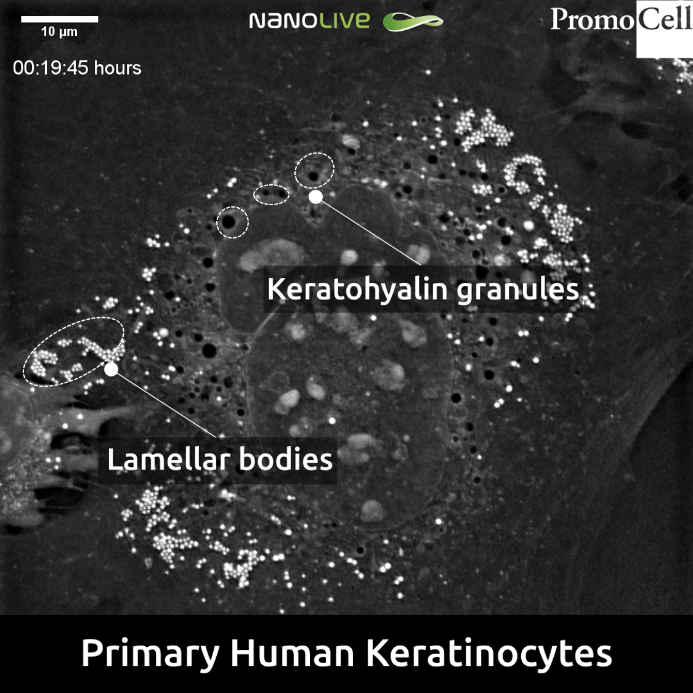
Figure 2. Keratohyalin granules and lamellar bodies in Primary Human Keratinocytes
The Nucleus
Unlike in our previous footages featuring NHEKs, in this sample the nucleus appears as a complex and highly compartmentalized organelle (Figure 3).
This phenomenon could maybe be explained by the effect of multiple divisions in the transition from the stratum basale to the stratum granulosum[7], [8], which has been known to include a number of asymmetric divisions mainly at the level of stratum basale that result in different cell fate and size[9], [10].
In fact, compared to the neighbour cell appearing in the lower left part of the footage, the cell at the centre seems to show an important increase in size.
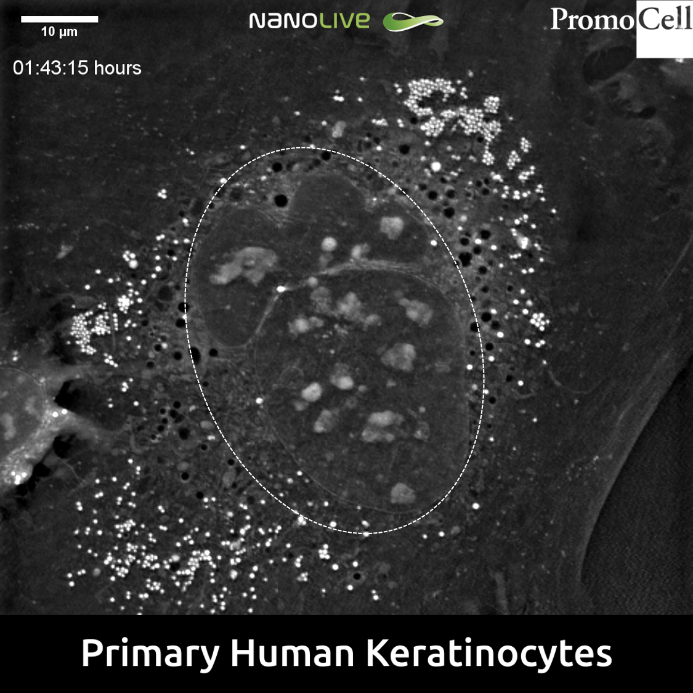
Figure 3. Complex and highly compartmentalized nucleus (dotted circle) in Primary Human Keratinocytes
Enjoy the full video here.
References
[1] J. A. McGrath, R. A. J. Eady, and F. M. Pope, “Anatomy and Organization of Human Skin,” in Rook’s Textbook of Dermatology, Malden, Massachusetts, USA: Blackwell Publishing, Inc., pp. 45–128.
[2] P. A. Kolarsick, M. Ann Kolarsick, and C. Goodwin, “Anatomy and Physiology of the Skin,” 2006.
[3] Practical immunodermatology. Springer, 2017.
[4] H. Yousef, M. Alhajj, and S. Sharma, Anatomy, Skin (Integument), Epidermis. StatPearls Publishing, 2019.
[5] A. Gedeon Matoltsy and M. N. Matoltsy, “THE CHEMICAL NATURE OF KERATOHYALIN GRANULES OF THE EPIDERMIS.”
[6] R. Joshi, “Learning from eponyms: George F. Odland and Odland bodies,” Indian Dermatol. Online J., vol. 5, no. 3, p. 334, Jul. 2014.
[7] “Polyploid cells and nuclei in keratinocyte primary cultures. (a-c)… | Download Scientific Diagram.” [Online]. Available: https://www.researchgate.net/figure/Polyploid-cells-and-nuclei-in-keratinocyte-primary-cultures-a-c-photographs-of_fig4_12400529. [Accessed: 27-Aug-2019].
[8] M. R. Gdula et al., “Remodeling of Three-Dimensional Organization of the Nucleus during Terminal Keratinocyte Differentiation in the Epidermis,” J. Invest. Dermatol., vol. 133, no. 9, pp. 2191–2201, Sep. 2013.
[9] M. I. Koster and D. R. Roop, “Asymmetric Cell Division in Skin Development: A New Look at an Old Observation,” Dev. Cell, vol. 9, no. 4, pp. 444–446, Oct. 2005.
[10] H.-Y. JIA et al., “Asymmetric stem-cell division ensures sustained keratinocyte hyperproliferation in psoriatic skin lesions,” Int. J. Mol. Med., vol. 37, no. 2, pp. 359–368, Feb. 2016.
Read our latest news
Revolutionizing lipid droplet analysis: insights from Nanolive’s Smart Lipid Droplet Assay Application Note
Introducing the Smart Lipid Droplet Assay: A breakthrough in label-free lipid droplet analysis Discover the power of Nanolive's Smart Lipid Droplet Assay (SLDA), the first smart digital assay to provide a push-button solution for analyzing lipid droplet dynamics,...
Food additives and gut health: new research from the University of Sydney
The team of Professor Wojciech Chrzanowski in the Sydney Pharmacy School at the University of Sydney have published their findings on the toxic effect of titanium nanoparticles found in food. The paper “Impact of nano-titanium dioxide extracted from food products on...
2023 scientific publications roundup
2023 has been a record year for clients using the Nanolive system in their scientific publications. The number of peer-reviewed publications has continued to increase, and there has been a real growth in groups publishing pre-prints to give a preview of their work....
Nanolive microscopes

3D CELL EXPLORER
Budget-friendly, easy-to-use, compact solution for high quality non-invasive 4D live cell imaging

3D CELL EXPLORER-fluo
Multimodal Complete Solution: combine high quality non-invasive 4D live cell imaging with fluorescence

CX-A
Automated live cell imaging: a unique walk-away solution for long-term live cell imaging of single cells and cell populations



